|
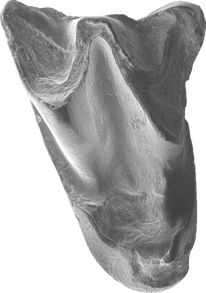
A frequent misconception about bats is that the oldest bats are known from the early Eocene (~52.5 million years ago) Green River Formation of western North America. In fact, there are earlier fragmentary bats known from Eocene deposits of both Australia and Portugal. Australonycteris clarkae and Archaeonycteris? praecursor both date to likely earliest Eocene sediments (1,2), while the complete skeletons of Icaronycteris index and Onychonycteris finneyi from the Green River Formation date to slightly younger late early Eocene deposits (3,4). In a new paper out today in Biology Letters, colleagues and I describe the first early Eocene bat from Asia, which is also—along with Australonycteris and Archaeonycteris? praecursor—among the oldest bats currently known (5). Bats actually occur nearly worldwide during the early Eocene (spanning 56-48 million years ago), being found in Africa, Australia, Europe, North America, South America, and the Indian subcontinent--which likely hadn’t completely collided with Asia at that point—by the end of the early Eocene (6). Until now, however, the oldest bats from mainland Asia dated to the middle Eocene ~43-44 million years ago. The new bat from the Junggar Basin of northwestern China, which we have named Altaynycteris aurora, likely dates to the first million years of the early Eocene based on biostratigraphic correlation with fossils from Inner Mongolia (5). This places Altaynycteris approximately contemporary with Australonycteris and Archaeonycteris? praecursor from Australia and Europe. Holotype upper first molar of Altaynycteris aurora. SEM photograph by Ni Xijun, IVPP. Finding early Eocene bats in central Asia is important, since we know several other groups of animals originated in Asia around this time period. The origin of both rodents and lagomorphs (rabbits, hares, and pikas) can confidently be traced to Asia, while many other groups of placental mammals have been hypothesized to originate there (7). Morphologically, Altaynycteris also appears primitive enough to represent a very early branch of the bat tree.
Only two teeth of Altaynycteris are currently known, but they possess enough similarity to other early Eocene bats (e.g., exaggerated W-shaped crests, lack of a mesostyle) that we are confident they belong to bats. These teeth, however, possess other similarities to non-bat insectivorous mammals from the Paleocene and early Eocene. They lack the common hook-shaped parastyle of most bats, and possess small cusps and crests (the metaconule and postparaconule crista) that are absent in most bats. The fact that Altaynycteris appears so primitive lends credence to the idea that bats may have a central Asian origin. 1) Hand, S., Novacek, M., Godthelp, H., & Archer, M. (1994). First Eocene bat from Australia. Journal of Vertebrate Paleontology, 14: 375-381. 2) Tabuce, R., Antunes, M.T., & Sigé, B. (2009). A new primitive bat from the earliest Eocene of Europe. Journal of Vertebrate Paleontology, 29: 627-630. 3) Jepsen, G.L. (1966). Early Eocene bat from Wyoming. Science, 154: 1333-1339. 4) Simmons, N.B., Seymour, K.L., Habersetzer, J., & Gunnell, G.F. (2008). Primitive early Eocene bat from Wyoming and the evolution of flight and echolocation. Nature, 451: 818-821. 5) Jones, M.F., Li, Q., Ni, X., & Beard, K.C. (2021). The earliest Asian bats (Mammalia: Chiroptera) address major gaps in bat evolution. Biology Letters, 17: 20210185. 6) Smith, T., Habersetzer, J., Simmons, N.B., & Gunnell, G.F. (2012). Systematics and paleobiogeography of early bats. In Evolutionary History of Bats: Fossils, Molecules, and Morphology (G.F. Gunnell & N.B. Simmons, eds.), 23-66. 7) Bowen, G.J., Clyde, W.C., Koch, P.L., Ting, S., Alroy, J., Tsubamoto, T., Wang, Y, & Wang, Y. (2002). Mammalian dispersal at the Paleocene/Eocene boundary. Science, 295: 2062-2065.
0 Comments
Powered flight—that is, the ability to remain aloft by flapping wings—has only evolved three times in vertebrates: once in mammals (bats), and twice in archosaurian reptiles (birds and pterosaurs). Major breakthroughs in bird origins occurred as long ago as the 1860s, with the discovery of Archaeopteryx and its mix of avian and reptilian features. Subsequent decades have reinforced the connection between birds and reptiles, and the past 30 years have introduced dozens of feathered theropods that each add a piece to the puzzle of bird origins. Pterosaurs and bats, however, have not had the same luck. The origins of both groups have remained some of the greatest mysteries in vertebrate paleontology. The first bats are known from the earliest Eocene (~55 Ma) and those with complete skeletons look effectively like modern bats (1). Similarly, the first pterosaurs date to the late Triassic (~220 Ma) and are fully flighted when they appear in the fossil record (2). Both groups are clearly very distinct from their closest relatives, although even those relatives have been debated. The only consensus being that bats belong within the mammalian superorder Laurasiatheria (including cows, whales, rhinos, hedgehogs, bears, and others) (3) while pterosaurs are archosaurs, likely closely related to dinosaurs (4, 5). There is hope on the horizon for pterosaurs, however, as a recent paper claims to have identified the closest fossil relatives of pterosaurs (6). A group of poorly-understood, nonflying Triassic reptiles known as lagerpetids are reconstructed as the sister-group to pterosaurs in a phylogenetic analysis based on new specimens and may shed light on the acquisition of features leading to pterosaurs and, in turn, to powered flight. The Brazilian lagerpetid Ixalerpeton. Art by Rodolfo Nogueira. Among the features shared by pterosaurs and lagerpetids are uniquely shaped semicircular canals of the inner ear. Several more distantly related living animals (including birds and primates) also possess enlarged anterior semicircular canals, which help to maintain equilibrium (6). Other endocranial and postcranial features also indicate the capacity for coordinated movements, and—although the authors only allude to this—may suggest an arboreal lifestyle for at least some lagerpetids.
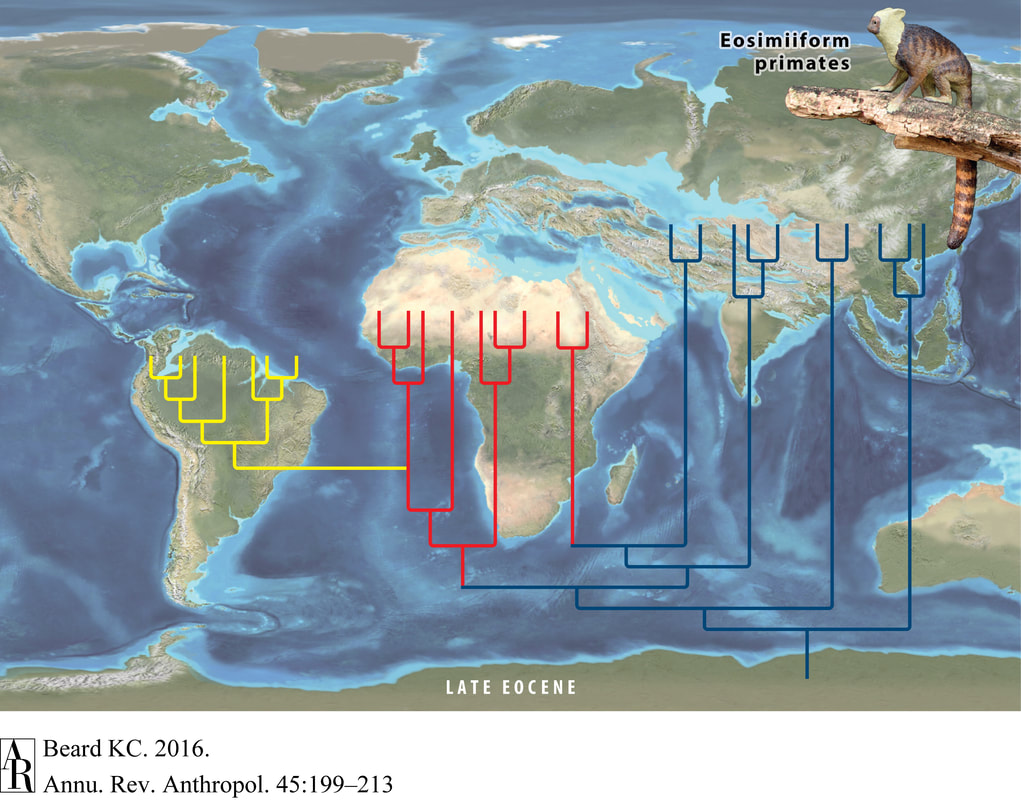
While lagerpetids may not represent the partially-flighted holy grail of pterosaur evolution, their recognition as close pterosaur relatives bridges many gaps in our understanding. That many adaptations indicate highly coordinated movement suggests that the most recent common ancestor of both pterosaurs and lagerpetids occupied a niche where these skills were necessary, perhaps as arboreal insectivores. Do these traits explain why the direct ancestors of pterosaurs have remained so elusive? We won’t be certain until we add more pieces to the puzzle, but at least now the picture is starting to take shape. Now, excuse me while I go to work on this bat mystery… References 1) Simmons, N.B., Seymour, K.L., Habersetzer, J., & Gunnell, G.F. (2008). Primitive early Eocene bat from Wyoming and the evolution of flight and echolocation. Nature, 451: 818-821. 2) Kellner, A.W.A. (2015). Comments on Triassic pterosaurs with discussion about ontogeny and description of new taxa. Anais da Academia Brasileira de Ciências, 87: 669-689. 3) Doronina, L., Churakov, G., Kuritzin, A., Shi, J., Baertsch, R., Clawson, H., & Schmitz, J. (2017). Speciation network in Laurasiatheria: retrophylogenomic signals. Genome Research, 27: 997-1003. 4) Nesbitt, S.J. & Hone, D.W.E. (2010). An external mandibular fenestra and other archosauriform character states in basal pterosaurs. Palaeodiversity, 3: 225-233. 5) Hone, D.W.E. (2012). Pterosaur research: recent advances and a future revolution. Acta Geologica Sinica, 86: 1366-1376. 6) Ezcurra, M.D., Nesbitt, S.J., Bronzati, M., Dalla Vecchia, F.M., Agnolin, F.L., Benson, R.B.J., Egli, F.B., et al. (2020). Enigmatic dinosaur precursors bridge the gap to the origin of Pterosauria. Nature, 588: 445-449. The presence of monkeys on both sides of the Atlantic Ocean (specifically, in both Africa and South America) has long puzzled biologists. The lineage of primates to which monkeys and apes belong--the anthropoids--is younger than the rifting of the Atlantic, so these animals could not have hitched a ride as the continents slowly drifted apart during the Mesozoic Era. No anthropoids are known in the North American fossil record until late in the Cenozoic, so dispersal from North to South America seems unlikely. As strange as it seems, the most likely dispersal route appears to be across the Atlantic Ocean. Perhaps even stranger, this trip almost certainly happened many times and crossed several oceans. A new paper by Seiffert et al. suggests that the trip across the Atlantic was accomplished by not one, but at least two different groups of monkeys (1). Modern South American monkeys belong to a lineage known as platyrrhines, and their closest living relatives are the Old World monkeys and apes, known as catarrhines. The new fossils described by Seiffert et al. belong to the Parapithecidae, a family of anthropoids known only from the fossil record which are more distantly related to both platyrrhines and catarrhines. Previously this group had only been found in Africa, so their presence in the early Oligocene of South America indicates a separate dispersal event across the Atlantic Ocean. South American monkeys are not the only group of mammals known to have crossed the Atlantic Ocean, other animals to make the trip include the endemic South American rodents of the group Caviomorpha (including guinea pigs, capybaras, and chinchillas). Like monkeys, the closest relatives of the caviomorphs--the hystricognath rodents--today live in Africa. What is even more interesting is that when all of these creatures set sail from Africa it too was an island continent, and the closest relatives of the anthropoids and hystricognath rodents were found in Asia. As improbable as it seems, these animals first crossed the Tethys Ocean from Asia to Africa and then in short order crossed the Atlantic Ocean from Africa to South America (2). Depiction of anthropoid colonization of Africa (red) and South America (yellow) from Beard (2016). Basal Eosimiiform anthropoids (Afrotarsiidae) and the common ancestor of platyrrhines and catarrhines colonized Africa independently. New findings by Seiffert et al. (2020) suggest a second independent colonization of South America. As is the case in South America, it seems like at least two groups of anthropoids colonized Africa. One, the Afrotarsiidae, forms one of the earliest branches of the anthropoid family tree and left no living descendents (3). The other group to colonize Africa would diversify into the ancestors of living catarrhines and platyrrhines, as well as a number of extinct groups including Parapithecidae. The pattern of multiple colonizations seems to be, perhaps by this point unsurprisingly, repeated in the hystricognath rodents (4).
The process by which the same groups of mammals can repeatedly cross oceans is illuminated by the documentation of large "floating islands" of vegetation that are launched out to sea following massive storms (see video). These natural rafts favor small, communal mammals that live near riverbanks. When the entire bank collapses, these animals set sail with the natural raft. Tens of thousands of rafts launching over millions of years nearly ensures that at least a few will contain viable colonies of primates and rodents. Evidence of an extinct group of monkeys that rafted from Africa to South America suggests that there may be even more ancient diversity waiting to be discovered on both continents. A solitary tooth of an extinct South American monkey named Perupithecus exhibits similarities to Talahpithecus, an African monkey belonging to the extinct family Oligopithecidae (5). If those two species are actually closely related, it may indicate a third independent crossing of the Atlantic, with potentially many more ocean crossings yet to be discovered. References 1) Seiffert, E.R., Tejedor, M.F., Fleagle, J.G., Novo, N.M., Cornejo, F.M., Bond, M., de Vries, D., & Campbell Jr., K.E. (2020). A parapithecid stem anthropoid of African origin in the Paleogene of South America. Science, 368: 194-197. 2) Beard, K.C. (2016). Out of Asia: anthropoid origins and the colonization of Africa. Annual Review of Anthropology, 45: 199-213. 3) Chaimanee, Y., Chavasseau, O., Beard, K.C., Kyaw, A.A., Soe, A.N., Sein, C., Lazzari, V., Marivaux, L., Marandat, B., Swe, M., Rugbumrung, M., Lwin, T., Valentin, X., Thein, Z.-M.-M., & Jaeger, J.-J. (2012). Late middle Eocene primate from Myanmar and the initial anthropoid colonization of Africa. PNAS, 109: 10293-10297. 4) Coster, P., Benammi, M., Lazzari, V., Billet, G., Martin, T., Salem, M., Bilal, A.A., Chaimanee, Y., Schuster, M., Valentin, X., Brunet, M., & Jaeger, J.-J. (2010). Gaudeamus lavocati sp. nov. (Rodentia, Hystricognathi) from the early Oligocene of Zallah, Libya: first African caviomorph?. Naturwissenschaften, 97: 697-706. 5) Bond, M., Tejedor, M.F., Campbell Jr., K.E., Chornogubsky, L., Novo, N., & Goin, F. (2015). Eocene primates of South America and the African origins of New World monkeys. Nature, 520: 538-541. A recent paper by Mazin and Poeuch in the journal Geobios describes the first known trackways of non-pterodactyloid (formerly called “Rhamphorynchoid”) pterosaurs (1). I began my master’s degree with the intention of studying pterosaur locomotion and trackways by using bats as living comparisons. “Rhamphorynchoid” pterosaurs – a hodgepodge group that contains all pterosaurs that don’t belong to the superfamily Pterodactyloidea – were particularly interesting to me due to the lack of known trackways and the amount of debate existing about how they walked. In the end, my interest in filling the gaps in our collective knowledge about how bats walk trumped my interest in non-pterodactyloid locomotion and I ended up abandoning that portion of my thesis. I remain interested in the terrestrial capabilities of pterosaurs, however, so I was very excited to read this new paper. Recognized trackways of pterodactyloid pterosaurs have become increasingly common since the 1990s (2), demonstrating that those late Jurassic to late Cretaceous animals were terrestrially adept quadrupeds with erect gaits. The absence of trackways belonging to the Triassic through Jurassic non-pterodactyloids, however, has sparked debate as to how terrestrially competent these animals were and whether they walked on two or four legs. This new paper describes trackways belonging to three new ichnospecies of non-pterodactyloid pterosaurs from an intertidal region in the late Jurassic Period of France. It suggests that non-pterodactyloids were not only quadrupedal on the ground, but that they held their hands with fingers aligned with the body, unlike pterodactyloids which held their fingers perpendicular. In 2015, Mark Witton presented a number of reasons that he felt suggested the terrestrial abilities of non-pterodactyloids were underestimated (3). Two of the points he focused on were whether the uropatagium (the part of the flight membrane that extends between the two hind legs) restricted movement of the legs, and whether the limbs were sprawled and how that would influence terrestrial locomotion. Witton observed that many living animals with extensive uropatagia are nonetheless adept walkers. My master’s research on living bats underscored this point as we documented walking and bounding gaits in the common vampire bat (Desmodus rotundus) and Seba’s short-tailed bat (Carollia perspicillata), while noting that other bats with relatively small patagia, like the greater sac-winged bat (Saccopteryx bilineata), appeared incapable of anything but a sprawling crawl (4). Other studies have shown bats with even larger uropatagia, such as the pallid bat (Antrozous pallidus), are capable of rapid and coordinated gaits (5). Walking postures of two non-pterodactyloid pterosaurs: sprawling gait of Dorygnathus (A); erect gait of Dimorphodon (B). Credit Mark Witton/PeerJ. Bats can inform Witton’s point about sprawling, too. Due to hindlimbs that are rotated 90⁰ from those of a typical mammal, bats cannot stand erect and must sprawl when on the ground. Their forelimbs--like those of pterosaurs--dwarf their hindlimbs, resulting in trackways in which the tracks produced by their hands are located outside those produced by the feet (4, 6). Nonetheless, Desmodus, Antrozous, and many other bat species are quite capable on the ground. Witton points out that not all non-pterodactlyoids appear to have had a sprawling posture, and those like Dimorphodon and Darwinopterus may have had terrestrial abilities similar to pterodactyloid pterosaurs. The trackways illustrated in the paper by Mazin and Poeuch seem to confirm this, as there is little evidence that the trackmaker possessed notably sprawled forelimbs.
Ultimately, it seems that the rarity of non-pterodactyloid pterosaur tracks is not a result of a lack of terrestrial ability, but may instead stem from differences in habitat and behavior. Not all environments are uniformly suited to preserve fossil trackways, and if non-pteryodactyloid pterosaurs were not occupying those favorable environments their trackways will not frequently be preserved. It is also unlikely that even the most terrestrially inept pterosaurs were completely helpless on the ground, and sprawling pterosaurs may have been able to employ a breaststrokelike crawl (similar to what we have documented in many phyllostomid bats and Saccopteryx). Ever since pterodactyoid tracks were first recognized in the fossil record they have been identified with increasing regularity all over the world. While non-pterodactyoid tracks may not prove to be as common as those of pterodactyoids, it is extremely likely that there are more out there, waiting to tell us more about how these animals moved and lived. References 1) Mazin, J.-M., & Pouech, J. (2020). The first non-pterodactyloid pterosaurian trackways and the terrestrial ability of non-pterodactyloid pterosaurs. Geobios https://doi.org/10.1016/j.geobios.2019.12.002 2) Lockley, M., Harris, J. D., & Mitchell, L. (2008). A global overview of pterosaur ichnology: tracksite distribution in space and time. Zitteliana, B28: 185-198. 3) Witton, M. P. (2015). Were early pterosaurs inept terrestrial locomotors? PeerJ, 3:e1018; DOI 10.7717/peerj.1018 4) Jones, M. F., & Hasiotis, S. T. (2018). Terrestrial behavior and trackway morphology of Neotropical bats. Acta Chiropterologica, 20: 229-250. 5) Dietz, C. L. (1973). Bat walking behavior. Journal of Mammalogy, 54: 790-792. 6) Lawrence, M. J. (1969). Some observations on non-volant locomotion in vespertilionid bats. Journal of Zoology, 157: 309-317. Throughout most of the world today, the major predators of insects that live under the bark of trees are birds--the avian woodpeckers of the family Picidae. This has not always been true, however, and it is not the case in modern Madagascar and Australia/New Guinea where the role of wood-boring insect predators are filled by mammals. These extant "mammalian woodpeckers" are the aye-aye, a lemur from Madagascar, and possums of the genus Dactylopsila, found in northern Australia and New Guinea. While avian woodpeckers use their bills to excavate holes in wood and use a long tongue to retrieve insects, these mammals use large teeth at the front of their mouths to excavate and elongated third or fourth fingers to grasp insects. Surprisingly, the "mammalian woodpecker" body plan is fairly common in the Cenozoic fossil record. The early placental mammal family Apatemyidae almost certainly occupied this niche, as evidenced by nearly complete skeletons of genera like Apatemys and Heterohyus, which display the same chisel-like front teeth and elongated fingers as living "mammalian woodpeckers" (1). Other groups of Cenozoic mammals are known only from cranial and dental remains but share enough features with living "mammal woodpeckers" that we can be fairly confident they also occupied this niche. These include the primitive marsupial Yalkiparodon (2), the African lemur-like primates Plesiopithecus and Propotto (3), and the early primate-like mammal Chiromyoides (4)*. Of course, as long as a niche exists, given enough time something will exploit it. In that case, perhaps it is not terribly surprising that mammal woodpeckers appear to have been relatively common in the early Cenozoic, but who was occupying the niche during the Mesozoic? Avian woodpeckers in the family Picidae had almost certainly not evolved yet (5), and we have no evidence for apatemyids or other "mammalian woodpeckers" at that time either. A paper published last week in Current Biology might provide some clues (6). In the paper, Xing et al. describe the foot of a Cretaceous bird preserved in Burmese amber with a surprisingly long third toe--a trait unknown in any other living or fossil bird. The authors speculate that this bird may have used its unusual toe for extracting insects, much like living aye-ayes. Artist reconstruction of Elektorornis chenguangi. Credit Zhongda Zhang/Current Biology. If this bird, Elektorornis chenguangi, did in fact use its elongated toe like an aye-aye it would represent another convergent evolution of the woodpecking mode of life. In an unexpected twist, this bird may have behaved more like one of the "mammalian woodpeckers" than the more closely related avian Picidae. Living avian woodpeckers possess a number of specialized traits to help them cope with the extreme stress caused by rapidly drumming their bills on wood (7). The suite of changes that was necessary for modern avian woodpeckers to evolve was almost certainly more complex than those necessary for mammalian woodpeckers, and perhaps the group of enantiornithine birds that Elektorornis belonged to also opted for the shorter evolutionary path. Of course, since we currently lack a skull or bill of Elektorornis this mode of life remains speculative, but the elongated toe is oddly reminiscent of the digits of aye-ayes and apatemyids. Although Picidae are the most familiar woodpeckers to the vast majority of us alive today, evolutionarily they may be the exception rather than the norm!
*Stay tuned for an upcoming paper about this bizarre little mammal References 1) Kenigswald, W. V., Rose, K. D., Grande, L., & Martin, R. D. (2005). First apatemyid skeleton from the lower Eocene Fossil Butte Member, Wyoming (USA), compared to the European apatemyid from Messel, Germany. Palaeontographica, Abteilung A: Palaozoologie - Stratigraphie, 272: 149-169. 2) Beck, R. M. D. (2009). Was the Oligo-Miocene Australian metatherian Yalkaparidon a 'mammalian woodpecker'? Biological Journal of the Linnean Society, 97: 1-17. 3) Gunnell, G. F., Boyer, D. M., Friscia, A. R., Heritage, S., Manthi, F. K., Miller, E. R., Sallam, H. M., Simmons, N. B., Stevens, N. J., & Seiffert, E. R. (2018). Fossil lemurs from Egypt and Kenya suggest an African origin for Madagascar's aye-aye. Nature Communications, 9: 1-12. 4) Szalay, F. S. & Delson, E. (1979). Evolutionary History of the Primates. Academic Press, New York, 580 pp. 5) Shakya, S. B., Fuchs, J., Pons, J.-M., & Sheldon, F. H. (2017). Tapping the woodpecker tree for evolutionary insight. Molecular Phylogenetics and Evolution, 116: 182-191. 6) Xing, L., O'Connor, J. K., Chiappe, L. M., McKellar, R. C., Carroll, N., Hu, H., Bai, M., & Lei, F. (2019). A new enantiornithine bird with unusual pedal proportions found in amber. Current Biology, 29: 1-6. 7) Wang, L., Cheung, J. T.-M., Pu, F., Li, D., Zhang, M., & Fan, Y. Why do woodpeckers resist head impact injury: a biomechanical investigation. PLOS One, 6: e26490. |
Matthew JonesMusings on evolution and paleontology-related research and news. Archives
June 2021
Categories
All
|



 RSS Feed
RSS Feed
